2013 SITAR Wiggins Award Winner Runner-up* Jenny M. Cundiff
*There was a tie
for runner up with Katherine Thomas whose submission was included in the spring
newsletter
 Jenny M. Cundiff
Jenny M. CundiffUniversity of Utah
Hierarchy and health: Physiological effects of interpersonal experiences associated with social status
Jenny M. Cundiff, Timothy W. Smith, Carolynne E. Baron, and Bert N. Uchino
University of Utah
Objective SES indicators or resources (e.g., education, income, occupation) predict health outcomes, but individuals’ perceptions of their social rank in relation to others (i.e., subjective social status) often predict morbidity and mortality after controlling for traditional indicators (Adler, et al., 2008). Hence, the association of social status with cardiovascular disease and other health outcomes may involve psychological and interpersonal factors (e.g., social comparison processes) associated with more frequent, pronounced, and prolonged physiological activation (e.g., increases in blood pressure, heart rate, neuroendocrine responses), which in turn contributes to the development of cardiovascular disease (Steptoe & Kivimaki, 2013).
Disentangling Relative Status, Dominance, and Threat
The experience of lower social standing during interpersonal interactions can reflect several factors that heighten physiological response. For example, lower perceived status in a potentially evaluative situation can lead to lower perceptions of relative ability and a greater perceived likelihood of failure or rejection. Such experiences have been previously linked to greater physiological stress response (Lam & Dickerson, 2013; Mendes, Blascovich, Major, & Seery, 2001). Additionally, low SES is also associated with greater self-reported exposure to dominant and controlling behavior from interaction partners in everyday life (Gallo, Smith, & Cox, 2006), and interactions with dominant (vs. deferential) partners evoke greater physiological stress responses (Newton, 2009; Smith et al., 2012).
The Present Study
Using a controlled social task involving a pre-recorded interaction partner, the current study examined the effects of manipulations of relative social status (i.e., family and personal income and education, and perceived social rank), dominant behavior of an interaction partner, and social-evaluative threat on cardiovascular and neuroendocrine stress responses. It was predicted that low participant relative status, dominant behavior by interaction partners, and high social-evaluative threat would independently evoke heightened physiological stress responses. The design also permitted examination of the additive versus synergistic effects of these status-related interpersonal processes, as well as a test of the general hypothesis that men are more responsive to status-related stressors than women (Newton, 2009). Perceptions of relative social status, partner behavior, state affect, and interaction goals were also assessed to evaluate the effectiveness of these experimental manipulations.
Method
Participants
Participants were 180 undergraduate men and women enrolled in the University of Utah’s Psychology Department research participation pool. Participants ranged in age from 17 to 60 years (median = 21 years), and 73.6% identified as Caucasian (10.7% Hispanic, 7.9% Asian American). Most were full-time students (81.4%), employed (60.3%), and made less than $15,000 annually (66.0%). Participants’ average subjective social status in the U.S. was 5.3 out of 10 rungs and average subjective social status in the community was 5.5 out of 10 rungs. Participants’ average parental income was between $75,000 and $125,000, average paternal education was a bachelor’s degree, and average maternal education was an associate’s degree.
Design and Procedures
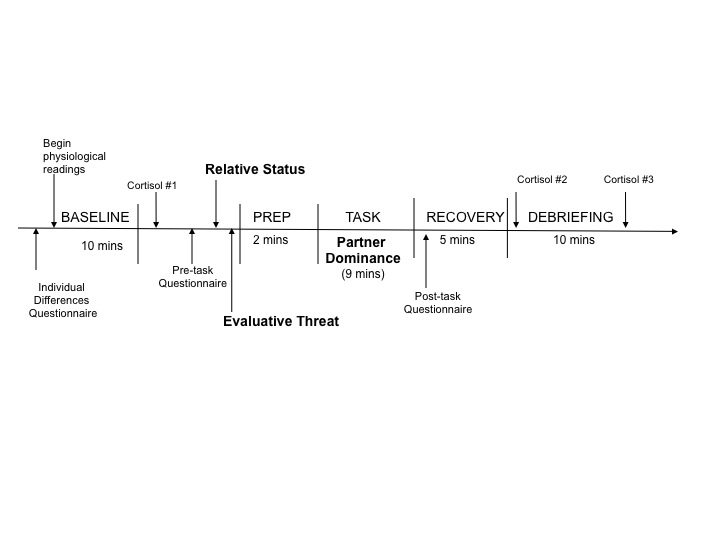
The study design was a 2 (Participant Relative Status: high vs. low) × 2 (Partner Dominance: high vs. low) × 2 (Evaluative Threat: high vs. low) × 2 (Sex: male vs. female) design. The figure below provides a timeline of study procedures.
Participant Relative Status manipulation. Participant Relative Status was manipulated by a supposed exchange of information between the participant and the interaction partner. That is, participants were presented with a demographic sheet of objective and subjective ratings of social status (identical in format to the one they completed earlier) and led to believe that these ratings were provided by their partner.
Social-Evaluative Threat manipulation. Low threat instructions specifically stated that neither the participants’ nor the partners’ responses during the interaction task would be evaluated, and asked only that participants speak clearly. High threat instructions stated that participants and their partners would be evaluated for competence, intelligence, skill, and accuracy of information. During the high threat condition, an experimenter sat and faced the participant, and supposedly made periodic ratings of both the participant and the partner.
Partner Dominance manipulation. The pre-recorded interaction partner was gender-matched with the participant, and the same male and female were recorded for all conditions. High dominant partners spoke in an assertive manner (i.e. confident and definitive), and low dominant partners spoke deferentially (i.e. hesitant speech, lack of certainty and confidence, questioning tone).
Interaction task. Participants “interacted” with the confederate recording in a series of three, 90-second exchanges on a currently debated topic. The topic about which the participant held the strongest opinion was chosen in all cases. In the pre-recorded remarks, the interaction partner always supported the opposing viewpoint.
Measures
Status.
Objective social status. We assessed income and education for the participant and his or her parents. Parental occupation was not assessed.
Subjective social status. In the background questionnaire participants also completed the MacArthur Scale of Subjective Social Status, consisting of two visual analogue scales in the form of ladders, one rating perceived status within the United States and one rating perceived status within the community, where community is defined by the rater (Adler et al., 2008). A visually identical ladder scale was used as a manipulation check following the interaction task.
Perceptions of confederate partner.
Partner behavior. As a manipulation check, a 32-item version of the Impact Message Inventory (IMI-C) (Schmidt, Wagner, & Kiesler, 1999) was used to assess participants’ perception of their partner’s interpersonal behavior.
Psychological reactions to the task.
State Affect. Anger and anxiety were assessed using the 12-item measure adapted from the State-Trait Personality Inventory (Spielberger, 1980)
Interpersonal goals during the interaction. The Circumplex Scale of Interpersonal Values (CSIV; Locke, 2000) is a self-report measure that asks participants to rate their goals. The original stem was adapted to read, “During the interaction…” to make it applicable to the current study, and four items were selected from the original eight items corresponding to each octant of the interpersonal circumplex, making sure that items were applicable to the interaction task (e.g., brief interaction with a stranger). This scale was reliable in the present sample (Cronbach’s alpha = .82).
Physiological reactions to the task.
Cardiovascular reactivity. Mean systolic blood pressure (SBP) and diastolic blood pressure (DBP) readings were calculated for each epoch to increase the reliability of these assessments (Kamarck et al., 1992). A Mindware 2000D Impedance Cardiograph was used along with spot electrodes placed in the tetra polar configuration (Thayer et al., 2008), and raw ECG data was visually inspected for artifacts and abnormal beats (Bernston, Quigley, Jane, & Boysen, 1990). HRV Analysis Software 5.2 (Mindware Cardiography system, Gahanna, Oh) was used to derive high frequency heart rate variability (hfHRV), and pre-ejection period (PEP) was calculated as the time interval in milliseconds (ms) between the Q-point of the ECG and the B-point of the dZ/dt signal.
Salivary cortisol. Given the typical timing of peak salivary cortisol levels in response to psychosocial stressors (Dickerson & Kemeny, 2004), samples were collected following baseline, and 25 and 35 minutes after the beginning of the interaction task. The two change scores (task minus baseline) were averaged and all analyses controlled time since waking.
Overview of Analyses
The results of 2 (Participant Relative Status: high or low) × 2 (Partner Dominance: high or low) × 2 (Evaluative Threat: high or low) × 2 (Sex: male or female) ANOVAs are presented below. Change scores were analyzed for affective and physiological responses to the task (Llabre, Spitzer, Saab, Ironson, & Schneiderman, 1991). Significant interactions were further analyzed using mean comparisons (Bernhardson, 1975).
Results
Summary of Manipulation Checks
The experimental manipulations were generally effective and specific. The Participant Relative Status manipulation had expected effects on participants’ SSS ladder ratings of themselves and their interaction partners, whereas the Partner Dominance and Evaluative Threat manipulations did not influence these ratings. The Partner Dominance manipulation had expected effects on the IMI-C ratings of the partner’s level of dominance and control during the interaction, whereas the Participant Relative Status and Evaluative Threat manipulations did not. Only the Evaluative Threat manipulation altered state anger, and to a lesser extent state anxiety, in the expected direction. High Evaluative Threat also resulted in higher reports of agentic or dominance goals during the interaction.
Physiological Reactivity during Social Interaction
Blood pressure response. SBP increased across study conditions overall, F(1,161) = 219.8, p < .001, η2 = .585 (M = 24.2 mm Hg). Additionally, the low Participant Relative Status group displayed larger increases than the high status group (M = 26.8 mmHg vs. 21.8 mmHg, SE = 1.1, 1.5), F(1,161) = 12.06, p<.001, η2 = .072. Also, the high Partner Dominance group displayed larger increases than did the low dominance group (M = 26.6 mmHg vs. 22.0 mmHg, SE = 1.1, 1.4), F(1,161) = 10.22, p<.001, η2 = .061. The Evaluative Threat effect approached significance, such that high threat participants tended to display larger increases than the low threat group (M = 25.5 mmHg vs. 22.7 mmHg, SE = 1.5, 1.2, respectively), F(1,161) = 3.65, p = .058, η2 = .023.
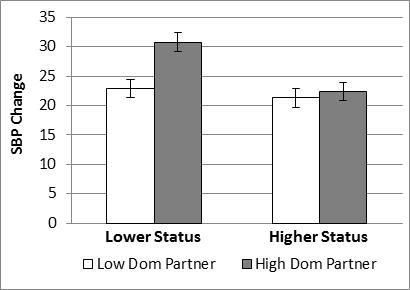
Results for SBP also revealed an interaction between Participant Relative Status and Partner Dominance, F(1,161) = 3.8, p = .05, η2 = .024 (see figure below).
Social-Evaluative Threat manipulation. Low threat instructions specifically stated that neither the participants’ nor the partners’ responses during the interaction task would be evaluated, and asked only that participants speak clearly. High threat instructions stated that participants and their partners would be evaluated for competence, intelligence, skill, and accuracy of information. During the high threat condition, an experimenter sat and faced the participant, and supposedly made periodic ratings of both the participant and the partner.
Partner Dominance manipulation. The pre-recorded interaction partner was gender-matched with the participant, and the same male and female were recorded for all conditions. High dominant partners spoke in an assertive manner (i.e. confident and definitive), and low dominant partners spoke deferentially (i.e. hesitant speech, lack of certainty and confidence, questioning tone).
Interaction task. Participants “interacted” with the confederate recording in a series of three, 90-second exchanges on a currently debated topic. The topic about which the participant held the strongest opinion was chosen in all cases. In the pre-recorded remarks, the interaction partner always supported the opposing viewpoint.
Measures
Status.
Objective social status. We assessed income and education for the participant and his or her parents. Parental occupation was not assessed.
Subjective social status. In the background questionnaire participants also completed the MacArthur Scale of Subjective Social Status, consisting of two visual analogue scales in the form of ladders, one rating perceived status within the United States and one rating perceived status within the community, where community is defined by the rater (Adler et al., 2008). A visually identical ladder scale was used as a manipulation check following the interaction task.
Perceptions of confederate partner.
Partner behavior. As a manipulation check, a 32-item version of the Impact Message Inventory (IMI-C) (Schmidt, Wagner, & Kiesler, 1999) was used to assess participants’ perception of their partner’s interpersonal behavior.
Psychological reactions to the task.
State Affect. Anger and anxiety were assessed using the 12-item measure adapted from the State-Trait Personality Inventory (Spielberger, 1980)
Interpersonal goals during the interaction. The Circumplex Scale of Interpersonal Values (CSIV; Locke, 2000) is a self-report measure that asks participants to rate their goals. The original stem was adapted to read, “During the interaction…” to make it applicable to the current study, and four items were selected from the original eight items corresponding to each octant of the interpersonal circumplex, making sure that items were applicable to the interaction task (e.g., brief interaction with a stranger). This scale was reliable in the present sample (Cronbach’s alpha = .82).
Physiological reactions to the task.
Cardiovascular reactivity. Mean systolic blood pressure (SBP) and diastolic blood pressure (DBP) readings were calculated for each epoch to increase the reliability of these assessments (Kamarck et al., 1992). A Mindware 2000D Impedance Cardiograph was used along with spot electrodes placed in the tetra polar configuration (Thayer et al., 2008), and raw ECG data was visually inspected for artifacts and abnormal beats (Bernston, Quigley, Jane, & Boysen, 1990). HRV Analysis Software 5.2 (Mindware Cardiography system, Gahanna, Oh) was used to derive high frequency heart rate variability (hfHRV), and pre-ejection period (PEP) was calculated as the time interval in milliseconds (ms) between the Q-point of the ECG and the B-point of the dZ/dt signal.
Salivary cortisol. Given the typical timing of peak salivary cortisol levels in response to psychosocial stressors (Dickerson & Kemeny, 2004), samples were collected following baseline, and 25 and 35 minutes after the beginning of the interaction task. The two change scores (task minus baseline) were averaged and all analyses controlled time since waking.
Overview of Analyses
The results of 2 (Participant Relative Status: high or low) × 2 (Partner Dominance: high or low) × 2 (Evaluative Threat: high or low) × 2 (Sex: male or female) ANOVAs are presented below. Change scores were analyzed for affective and physiological responses to the task (Llabre, Spitzer, Saab, Ironson, & Schneiderman, 1991). Significant interactions were further analyzed using mean comparisons (Bernhardson, 1975).
Results
Summary of Manipulation Checks
The experimental manipulations were generally effective and specific. The Participant Relative Status manipulation had expected effects on participants’ SSS ladder ratings of themselves and their interaction partners, whereas the Partner Dominance and Evaluative Threat manipulations did not influence these ratings. The Partner Dominance manipulation had expected effects on the IMI-C ratings of the partner’s level of dominance and control during the interaction, whereas the Participant Relative Status and Evaluative Threat manipulations did not. Only the Evaluative Threat manipulation altered state anger, and to a lesser extent state anxiety, in the expected direction. High Evaluative Threat also resulted in higher reports of agentic or dominance goals during the interaction.
Physiological Reactivity during Social Interaction
Blood pressure response. SBP increased across study conditions overall, F(1,161) = 219.8, p < .001, η2 = .585 (M = 24.2 mm Hg). Additionally, the low Participant Relative Status group displayed larger increases than the high status group (M = 26.8 mmHg vs. 21.8 mmHg, SE = 1.1, 1.5), F(1,161) = 12.06, p<.001, η2 = .072. Also, the high Partner Dominance group displayed larger increases than did the low dominance group (M = 26.6 mmHg vs. 22.0 mmHg, SE = 1.1, 1.4), F(1,161) = 10.22, p<.001, η2 = .061. The Evaluative Threat effect approached significance, such that high threat participants tended to display larger increases than the low threat group (M = 25.5 mmHg vs. 22.7 mmHg, SE = 1.5, 1.2, respectively), F(1,161) = 3.65, p = .058, η2 = .023.
Results for SBP also revealed an interaction between Participant Relative Status and Partner Dominance, F(1,161) = 3.8, p = .05, η2 = .024 (see figure below).
In sum, the main effects of Participant Relative Status and Partner Dominance were largely attributable to the particularly large increase in SBP associated with the combination of lower relative status and interacting with a high dominant partner.
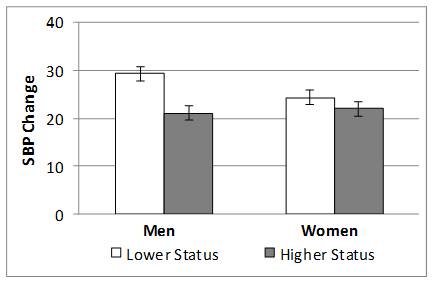
There was also an interaction between Participant Relative Status and Sex on SBP, F(1,161) = 4.0, p = .048 η2 = .025(see figure below). Mean comparisons revealed that lower relative status evoked greater SBP increases for men but not women, t(161) = 2.4 , p = .008.
There was also an interaction between Participant Relative Status and Sex on SBP, F(1,161) = 4.0, p = .048 η2 = .025(see figure below). Mean comparisons revealed that lower relative status evoked greater SBP increases for men but not women, t(161) = 2.4 , p = .008.
DBP also increased across study conditions overall, F(1,161) = 182.8, p < .001, η2 = .54 (M = 12.6 mm Hg). Only the Participant Relative Status main effect was significant, such that lower status participants showed significantly larger increases in DBP during the task (M = 14.4 mmHg vs. 11.1 mmHg, SE = .62, .84), F(1,161) = 15.87, p <.001, η2 = .092.
Heart Rate, Pre-Ejection period, and high frequency Heart Rate Variability. HR increased across study conditions overall, F(1,161) = 52.85, p < .001, η2 = .253 (M = 14.05 bpm). There was no main effect of Participant Relative Status, but both Partner Dominance and Evaluative Threat significantly affected changes in HR. Participants with high dominant partners had significantly greater increases in HR than did participants with low dominant partners (M = 18.9 bpm vs. 9.8 bpm, SE = 1.2,1.7), F(1,161) = 25.74, p<.001, η2 = .142, and those in the high Evaluative Threat group displayed greater HR responses than those in the low threat group (M = 15.7 bpm vs. 12.2 bpm, SE = 1.7, 1.2), F(1,161) = 5.79, p = .017, η2 = .036.
There was a large decrease in PEP across study conditions overall, F(1,129) = 118.90, p < .001, η2 = .480 (M = -16.6 msec), indicating increased cardiac sympathetic activity during the task. There was no main effect of either Participant Relative Status or Evaluative Threat on change in PEP. However, there was a main effect of Partner Dominance, such that interacting with a high dominant partner evoked greater decreases in PEP than did low dominant partners (M = -18.0 msec vs. -15.4 msec, SE = 1.2, 1.5), F(1,129) = 5.21, p = .02, η2 = .039. Further, the main effect of Partner Dominance was further qualified by a significant interaction with sex, F(1,129) = 4.15, p = .044, η2 = .031. Only men showed larger decreases in PEP response to a dominant partner, t(129) = 2.2, p = .015.
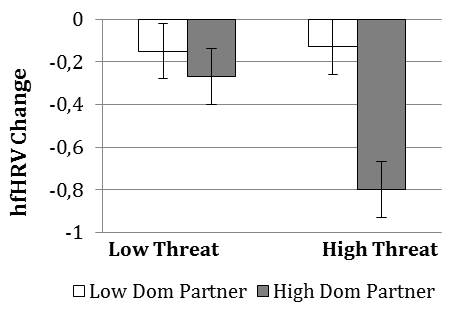
Lastly, there was a significant decrease in hfHRV across study conditions overall, F(1,150) = 6.99, p = .009, η2 = .045 (M = -.36), indicating parasympathetic withdrawal during the task. There was no main effect of Participant Relative Status on change in hfHRV. However, there was a main effect of Partner Dominance; high (vs. low) partner dominance resulted in larger decreases in hfHRV (M = -.53 vs. -.21, SE = .10, .13), F(1, 150) = 8.07, p = .005, η2 = .051. Also, high Evaluative Threat participants tended to display greater decreases in hfHRV than those in the low threat group (M = -.49 vs. -.21, SE = .13, .10), but this difference only approached significance, F(1,150) = 3.37, p = .068, η2 = .022. These main effects were further qualified by a Partner Dominance x Evaluative Threat interaction, F(1,150) = 3.98, p = .048, η2 = .026 (see figure below).
Heart Rate, Pre-Ejection period, and high frequency Heart Rate Variability. HR increased across study conditions overall, F(1,161) = 52.85, p < .001, η2 = .253 (M = 14.05 bpm). There was no main effect of Participant Relative Status, but both Partner Dominance and Evaluative Threat significantly affected changes in HR. Participants with high dominant partners had significantly greater increases in HR than did participants with low dominant partners (M = 18.9 bpm vs. 9.8 bpm, SE = 1.2,1.7), F(1,161) = 25.74, p<.001, η2 = .142, and those in the high Evaluative Threat group displayed greater HR responses than those in the low threat group (M = 15.7 bpm vs. 12.2 bpm, SE = 1.7, 1.2), F(1,161) = 5.79, p = .017, η2 = .036.
There was a large decrease in PEP across study conditions overall, F(1,129) = 118.90, p < .001, η2 = .480 (M = -16.6 msec), indicating increased cardiac sympathetic activity during the task. There was no main effect of either Participant Relative Status or Evaluative Threat on change in PEP. However, there was a main effect of Partner Dominance, such that interacting with a high dominant partner evoked greater decreases in PEP than did low dominant partners (M = -18.0 msec vs. -15.4 msec, SE = 1.2, 1.5), F(1,129) = 5.21, p = .02, η2 = .039. Further, the main effect of Partner Dominance was further qualified by a significant interaction with sex, F(1,129) = 4.15, p = .044, η2 = .031. Only men showed larger decreases in PEP response to a dominant partner, t(129) = 2.2, p = .015.
Lastly, there was a significant decrease in hfHRV across study conditions overall, F(1,150) = 6.99, p = .009, η2 = .045 (M = -.36), indicating parasympathetic withdrawal during the task. There was no main effect of Participant Relative Status on change in hfHRV. However, there was a main effect of Partner Dominance; high (vs. low) partner dominance resulted in larger decreases in hfHRV (M = -.53 vs. -.21, SE = .10, .13), F(1, 150) = 8.07, p = .005, η2 = .051. Also, high Evaluative Threat participants tended to display greater decreases in hfHRV than those in the low threat group (M = -.49 vs. -.21, SE = .13, .10), but this difference only approached significance, F(1,150) = 3.37, p = .068, η2 = .022. These main effects were further qualified by a Partner Dominance x Evaluative Threat interaction, F(1,150) = 3.98, p = .048, η2 = .026 (see figure below).
High (vs. low) Evaluative Threat resulted in decreased hfHRV only when interacting with a high dominant partner, t(150) = 2.79 , p = .003, and interacting with a high (vs. low) dominant partner resulted in decreased hfHRV only in the high Evaluative Threat group, t(150) = 3.53 , p < .001.
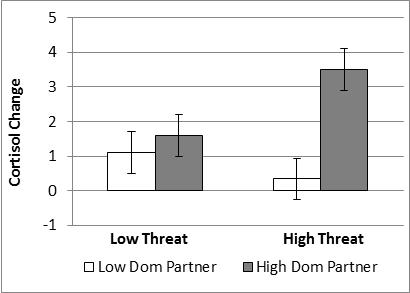
Salivary cortisol. On average, participants across conditions overall showed a significant increase in salivary cortisol, F(1,154) = 7.1, p < .01, η2 = .046 (M = 1.7 nmol/L). There were no main effects of manipulated variables. However, there was a significant Partner Dominance x Evaluative Threat interaction , F(1,154) = 4.0, p = .047, η2 = .026 (see figure below), which was similar to the interaction of these manipulations on hfHRV.
Salivary cortisol. On average, participants across conditions overall showed a significant increase in salivary cortisol, F(1,154) = 7.1, p < .01, η2 = .046 (M = 1.7 nmol/L). There were no main effects of manipulated variables. However, there was a significant Partner Dominance x Evaluative Threat interaction , F(1,154) = 4.0, p = .047, η2 = .026 (see figure below), which was similar to the interaction of these manipulations on hfHRV.
High (vs. low) Evaluative Threat resulted in greater cortisol reactivity when participants interacted with a high dominant partner, t(154) = 2.0, p = .02, but not with a low dominant partner. Further, interacting with a high (vs. low) dominant partner resulted in greater cortisol reactivity in the high Evaluative Threat group, t(154) = 3.33, p < .001 , but not in the low threat group.
Cardiovascular Recovery
Across conditions overall, participants showed increased SBP (M = 9.26 mm Hg), F(1,161) = 259.67, p < .001,η2 = .623, DBP (M = 3.22 mm Hg), F(1,161) = 61.30, p < .001 ,η2 = .281, and HR (M = 2.80 bpm), F(1,161) = 42.48 ,p < .001, η2 = .215, as well as decreased PEP (M = -8.56 msec), F(1,129) = 134.83 , p < .001 , η2 = .511 during recovery compared to baseline. For main effects in the main analyses, there was only a small effect of Participant Relative Status on DBP recovery, F(1,161) = 4.0, p = .047, η2 = .025, such that lower status resulted in higher levels of sustained DBP reactivity.
For both SBP and DBP there was a significant interaction between Participant Relative Status and Sex, F(1,161) = 6.80, p = .01, η2 = .042, and F(1,161) = 5.67, p = .035, η2 = .035, respectively. In both cases, mean comparisons revealed that only men showed an effect of Participant Relative Status on blood pressure recovery, both t(161) > 2.4, p < .01 .
Discussion
To experimentally test the effects of aspects of SES on physiological reactivity, the current study manipulated three features of social context related to social hierarchy – social rank or status relative to an interaction partner, the partner’s degree of dominant behavior, and the presence of social-evaluative threat. The experimental manipulations were generally effective, with expected effects on ratings of subjective social status, perceptions of partner behavior, hierarchy-related goals, and changes in negative affect. Further, the interaction task evoked substantial cardiovascular and neuroendocrine responses, as well as increases in negative affect. Hence, this experimental analogue provides a reasonable test of the effects of aspects of social status on physiological reactivity during social interaction.
Overall, results support the viability of multiple interpersonal pathways from low social status to psychophysiological processes related to disease development. Lower perceived relative status resulted in greater increases in SBP and DBP during social interaction, as well as sustained increases in DBP following the task. Interacting with a more dominant partner, an experience that is more common for individuals lower in SES (Gallo, et al., 2006), resulted in greater increases in SBP and HR, as well as larger decreases in PEP and hfHRV. These latter effects suggest that interactions with a more dominant partner evoke both greater sympathetic activation and greater withdrawal of parasympathetic activity. Finally, higher levels of social-evaluative threat evoked larger increases in HR and marginally larger increases in SBP.
These aspects of the low status social context also had synergistic effects on physiological responses in several instances. For example, high levels of partner dominance combined with low relative social status produced particularly large increases in SBP, and the combination of high evaluative threat and dominant partner behavior produced particularly large decreases in parasympathetic activity and large increases in salivary cortisol. Hence, both lower relative status and high evaluative threat appeared to increase the stress of interacting with a high dominant partner. Similarly, interacting with a more dominant partner may increase the stress associated with social-evaluative contexts and lower relative social status. Thus, some of the unhealthy physiological effects of low social status may result from combinations of social experiences that are more common in low status contexts.
In several instances, these aspects of social status had different effects on men and women. For example, lower relative status evoked larger increases in SBP among men but not women, a pattern that continued into the recovery period. Further, only men showed greater decreases in PEP in response to dominant interaction partners. These results support prior research in which men are more physiologically responsive to social hierarchy cues (Newton, 2009).
Conclusions
The current study provides evidence that interpersonal interactions and experiences may contribute to the association between SES and cardiovascular health through the mechanism of physiological activation. To the extent that these controlled laboratory-based interactions resemble interpersonal interactions in daily life, recurring patterns of everyday social experiences (e.g., exposure to social dominance expressed by others, perceptions of low relative status) may be an important pathway linking the broader social context to cardiovascular disease in individual persons (Smith et al., 2012). Additionally, these commonly co-occurring aspects of low SES social contexts appear to combine synergistically to evoke particularly large physiological responses. Hence, it may be important to consider multiple aspects of the social context associated with social hierarchy (e.g. relative rank and exposure to dominance), in order to explicate the effects of social status on physiological mechanisms and ultimately on disease.
References
Adler, N. E. (2009). Health disparities through a psychological lens. American Psychologist, 64(8), 663-673.
Adler, N. E., Singh-Manoux, A., Schwartz, J., Stewart, J., Matthews, K., & Marmot, M. (2008). Social status and health: A comparison of British civil servants in Whitehall-II with European- and African-Americans in CARDIA. Social Science & Medicine, 66(5), 1034-1045.
Almeida, D. M., Neupert, S. D., Banks, S. R., & Serido, J. (2005). Do daily stress processes account for socioeconomic health disparities? The Journals of Gerontology: Series B: Psychological Sciences and Social Sciences, 60B(2,SpecIssue), 34-39.
Bernhardson, C. S. (1975). 375: Type I error rates when multiple comparison procedures follow a significant F test of ANOVA. Biometrics, 229-232.
Berntson, G. G., Quigley, K. S., Jang, J., and Boysen, S. (1990). An approach to artifact identification: Application to heart period data.Psychophysiology 27: 586–598.
Cacioppo, J. T., & Petty, R. E. (1983). Social psychophysiology: A sourcebook. New York: Guildford Press.
Chida, Y., & Steptoe, A. (2010). Greater Cardiovascular Responses to Laboratory Mental Stress Are Associated With Poor Subsequent Cardiovascular Risk Status: A Meta-Analysis of Prospective Evidence. Hypertension, 55(4), 1026-1032.
Cundiff, J.M., Smith, T.W., Uchino, B.N., & Berg, C.A. (2012). Subjective Social Status: Construct Validity and Associations with Psychosocial Vulnerability and Self-Rated Health. International Journal of Behavioral Medicine, 20(1), 148-158.
Cundiff, J.M., Uchino, B.N., Smith, T.W., & Birmingham, W. (in press). Socioeconomic status and health: Education and income are independent and joint predicotrs of ambulatory blood pressure. Journal of Behavioral Medicine.
Dickerson, S. S., & Kemeny, M. E. (2004). Acute stressors and cortisol responses: a theoretical integration and synthesis of laboratory research. Psychological Bulletin, 130(3), 355-391.
Gallo, L. C., Smith, T. W., & Cox, C. M. (2006). Socioeconomic Status, Psychosocial Processes, and Perceived Health: An Interpersonal Perspective. Annals of Behavioral Medicine, 31(2), 109-119.
Hamer, M., Endrighi, R., Venuraju, S. M., Lahiri, A., & Steptoe, A. (2012). Cortisol responses to mental stress and the progression of coronary artery calcification in healthy men and women. PloS one, 7(2), e31356.
Jennings, J. R., Kamarck, T., Stewart, C., Eddy, M., & Johnson, P. (1992). Alternate cardiovascular baseline assessment techniques: Vanilla or resting baseline. Psychophysiology, 29(6), 742-750.
Kamarck, T. W., Jennings, J. R., Debski, T. T., Glickman‐Weiss, E., Johnson, P. S., Eddy, M. J., & Manuck, S. B. (1992). Reliable Measures of Behaviorally‐Evoked Cardiovascular Reactivity from a PC‐Based Test Battery: Results from Student and Community Samples. Psychophysiology, 29(1), 17-28.
Lam, S., & Dickerson, S.S. (2013). Social relationships, social threat, and health. In M.L. Neuman & N.A. Roberts (Eds), Health and social relationships: The good, the bad, and the complicated (pp. 19-38). Washington, DC: American Psychological Association.
Llabre, M. M., Spitzer, S. B., Saab, P. G., Ironson, G. H., & Schneiderman, N. (1991). The Reliability and Specificity of Delta Versus Residualized Change as Measures of Cardiovascular Reactivity to Behavioral Challenges. Psychophysiology, 28(6), 701 - 711.
Locke, K. D. (2000). Circumplex scales of interpersonal values: Reliability, validity, and applicability to interpersonal problems and personality disorders. Journal of Personality Assessment, 75(2), 249-267.
Matthews, K., Gallo, L. C., & Taylor, S. E. (2010). Are psychosocial factors mediators of socioeconomic satus and health connections? A progress report and blueprint for the future. Annals of the New York Acadamy of Sciences, 1186, 146-173.
Mendelson, T., Thurston, R. C., & Kubzansky, L. D. (2008). Affective and cardiovascular effects of experimentally-induced social status. Health Psychology, 27(4), 482-489.
Mendes, W. B., Blascovich, I., Major, B., & Seery, M. D. (2001). Challenge and threat responses during downward and upward social comparisons. European Journal of Social Psychology, 31, 477-497.
Miller, R., Plessow, F., Rauh, M., Groschl, M., & Kirschbaum, C. (2013). Comparison of salivary cortisol as measured by different immunoassays and tandem mass spectrometry. Psychoneuroendocrinology, 38, 50-57.
Nealey-Moore, J. B., Smith, T. W., Uchino, B. N., Hawkins, M. W., & Olson-Cerny, C. (2007). Cardiovascular reactivity during positive and negative marital interactions. Journal of Behavioral Medicine, 30, 505-519.
Newton, T. L. (2009). Cardiovascular functioning, personality, and the social world: the domain of hierarchical power. Neuroscience and Biobehavioral Reviews, 33(2), 145-159.
Operario, D., Adler, N. E., & Williams, D. R. (2004). Subjective social status: Reliability and predictive utility for global health. Psychology & Health, 19, 237-246.
Scheepers, D. (2009). Turning social identity threat into challenge: Status stability and cardiovascular reactivity during intergroup competition. Journal of Experimental Social Psychology, 45, 228 - 233.
Schmidt, J. A., Wagner, C. C., & Kiesler, D. J. (1999). Psychometric and circumplex properties of the octant scale Impact Message Inventory (IMI-C): A structural evaluation. Journal of Counseling Psychology, 46(3), 325-334.
Smith, T.W., & Christensen, A.J. (1992). Cardiovascular reactivity and interpersonal relations: Psychosomatic processes in social context. Journal of Social and Clinical Psychology, 11, 279-301.
Smith, T. W., Cundiff, J. M., & Uchino, B. N. (2012). Interpersonal motives and cardiovascular response: Mechanisms linking dominance and social status with cardiovascular disease. In R. A. Wright & G. H. E. Gendolla (Eds.), How motivation affects cardiovascular response: Mechanisms and applications. (pp. 287-305). Washington, DC US: American Psychological Association.
Smith, T. W., Ruiz, J. M., & Uchino, B. N. (2000). Vigilance, active coping, and cardiovascular reactivity during social interaction in young men. Health Psychology, 19(4), 382-392.
Steptoe, A., & Kivimaki, M. (2013). Stress and cardiovascular disease: An update on current knowledge. Annual Review of Public Health, 34, 337-354.
Spielberger, C. D. (1980). Preliminary manual for the State-Trait Personality Inventory. Tampa: University of South Florida, Human Resources Institute.
Thayer, J. F., Hansen, A. L., & Johnson, B. D. (2008). Non-invasive assessment of autonomic influences on the heart: Impedance cardiography and heart rate variability. In L. Leuken & L. Gallo (Eds.), Handbook of physiological research methods in health psychology (pp. 183-209). Thousand Oaks, CA: Sage.
Wright, R. A., Killebrew, K., & Pimpalapure, D. (2002). Cardiovascular incentive effects where a challenge is unfixed: Demonstrations involving social evaluation, evaluator status, and monetary reward. Psychophysiology, 39(2), 188-197.
Cardiovascular Recovery
Across conditions overall, participants showed increased SBP (M = 9.26 mm Hg), F(1,161) = 259.67, p < .001,η2 = .623, DBP (M = 3.22 mm Hg), F(1,161) = 61.30, p < .001 ,η2 = .281, and HR (M = 2.80 bpm), F(1,161) = 42.48 ,p < .001, η2 = .215, as well as decreased PEP (M = -8.56 msec), F(1,129) = 134.83 , p < .001 , η2 = .511 during recovery compared to baseline. For main effects in the main analyses, there was only a small effect of Participant Relative Status on DBP recovery, F(1,161) = 4.0, p = .047, η2 = .025, such that lower status resulted in higher levels of sustained DBP reactivity.
For both SBP and DBP there was a significant interaction between Participant Relative Status and Sex, F(1,161) = 6.80, p = .01, η2 = .042, and F(1,161) = 5.67, p = .035, η2 = .035, respectively. In both cases, mean comparisons revealed that only men showed an effect of Participant Relative Status on blood pressure recovery, both t(161) > 2.4, p < .01 .
Discussion
To experimentally test the effects of aspects of SES on physiological reactivity, the current study manipulated three features of social context related to social hierarchy – social rank or status relative to an interaction partner, the partner’s degree of dominant behavior, and the presence of social-evaluative threat. The experimental manipulations were generally effective, with expected effects on ratings of subjective social status, perceptions of partner behavior, hierarchy-related goals, and changes in negative affect. Further, the interaction task evoked substantial cardiovascular and neuroendocrine responses, as well as increases in negative affect. Hence, this experimental analogue provides a reasonable test of the effects of aspects of social status on physiological reactivity during social interaction.
Overall, results support the viability of multiple interpersonal pathways from low social status to psychophysiological processes related to disease development. Lower perceived relative status resulted in greater increases in SBP and DBP during social interaction, as well as sustained increases in DBP following the task. Interacting with a more dominant partner, an experience that is more common for individuals lower in SES (Gallo, et al., 2006), resulted in greater increases in SBP and HR, as well as larger decreases in PEP and hfHRV. These latter effects suggest that interactions with a more dominant partner evoke both greater sympathetic activation and greater withdrawal of parasympathetic activity. Finally, higher levels of social-evaluative threat evoked larger increases in HR and marginally larger increases in SBP.
These aspects of the low status social context also had synergistic effects on physiological responses in several instances. For example, high levels of partner dominance combined with low relative social status produced particularly large increases in SBP, and the combination of high evaluative threat and dominant partner behavior produced particularly large decreases in parasympathetic activity and large increases in salivary cortisol. Hence, both lower relative status and high evaluative threat appeared to increase the stress of interacting with a high dominant partner. Similarly, interacting with a more dominant partner may increase the stress associated with social-evaluative contexts and lower relative social status. Thus, some of the unhealthy physiological effects of low social status may result from combinations of social experiences that are more common in low status contexts.
In several instances, these aspects of social status had different effects on men and women. For example, lower relative status evoked larger increases in SBP among men but not women, a pattern that continued into the recovery period. Further, only men showed greater decreases in PEP in response to dominant interaction partners. These results support prior research in which men are more physiologically responsive to social hierarchy cues (Newton, 2009).
Conclusions
The current study provides evidence that interpersonal interactions and experiences may contribute to the association between SES and cardiovascular health through the mechanism of physiological activation. To the extent that these controlled laboratory-based interactions resemble interpersonal interactions in daily life, recurring patterns of everyday social experiences (e.g., exposure to social dominance expressed by others, perceptions of low relative status) may be an important pathway linking the broader social context to cardiovascular disease in individual persons (Smith et al., 2012). Additionally, these commonly co-occurring aspects of low SES social contexts appear to combine synergistically to evoke particularly large physiological responses. Hence, it may be important to consider multiple aspects of the social context associated with social hierarchy (e.g. relative rank and exposure to dominance), in order to explicate the effects of social status on physiological mechanisms and ultimately on disease.
References
Adler, N. E. (2009). Health disparities through a psychological lens. American Psychologist, 64(8), 663-673.
Adler, N. E., Singh-Manoux, A., Schwartz, J., Stewart, J., Matthews, K., & Marmot, M. (2008). Social status and health: A comparison of British civil servants in Whitehall-II with European- and African-Americans in CARDIA. Social Science & Medicine, 66(5), 1034-1045.
Almeida, D. M., Neupert, S. D., Banks, S. R., & Serido, J. (2005). Do daily stress processes account for socioeconomic health disparities? The Journals of Gerontology: Series B: Psychological Sciences and Social Sciences, 60B(2,SpecIssue), 34-39.
Bernhardson, C. S. (1975). 375: Type I error rates when multiple comparison procedures follow a significant F test of ANOVA. Biometrics, 229-232.
Berntson, G. G., Quigley, K. S., Jang, J., and Boysen, S. (1990). An approach to artifact identification: Application to heart period data.Psychophysiology 27: 586–598.
Cacioppo, J. T., & Petty, R. E. (1983). Social psychophysiology: A sourcebook. New York: Guildford Press.
Chida, Y., & Steptoe, A. (2010). Greater Cardiovascular Responses to Laboratory Mental Stress Are Associated With Poor Subsequent Cardiovascular Risk Status: A Meta-Analysis of Prospective Evidence. Hypertension, 55(4), 1026-1032.
Cundiff, J.M., Smith, T.W., Uchino, B.N., & Berg, C.A. (2012). Subjective Social Status: Construct Validity and Associations with Psychosocial Vulnerability and Self-Rated Health. International Journal of Behavioral Medicine, 20(1), 148-158.
Cundiff, J.M., Uchino, B.N., Smith, T.W., & Birmingham, W. (in press). Socioeconomic status and health: Education and income are independent and joint predicotrs of ambulatory blood pressure. Journal of Behavioral Medicine.
Dickerson, S. S., & Kemeny, M. E. (2004). Acute stressors and cortisol responses: a theoretical integration and synthesis of laboratory research. Psychological Bulletin, 130(3), 355-391.
Gallo, L. C., Smith, T. W., & Cox, C. M. (2006). Socioeconomic Status, Psychosocial Processes, and Perceived Health: An Interpersonal Perspective. Annals of Behavioral Medicine, 31(2), 109-119.
Hamer, M., Endrighi, R., Venuraju, S. M., Lahiri, A., & Steptoe, A. (2012). Cortisol responses to mental stress and the progression of coronary artery calcification in healthy men and women. PloS one, 7(2), e31356.
Jennings, J. R., Kamarck, T., Stewart, C., Eddy, M., & Johnson, P. (1992). Alternate cardiovascular baseline assessment techniques: Vanilla or resting baseline. Psychophysiology, 29(6), 742-750.
Kamarck, T. W., Jennings, J. R., Debski, T. T., Glickman‐Weiss, E., Johnson, P. S., Eddy, M. J., & Manuck, S. B. (1992). Reliable Measures of Behaviorally‐Evoked Cardiovascular Reactivity from a PC‐Based Test Battery: Results from Student and Community Samples. Psychophysiology, 29(1), 17-28.
Lam, S., & Dickerson, S.S. (2013). Social relationships, social threat, and health. In M.L. Neuman & N.A. Roberts (Eds), Health and social relationships: The good, the bad, and the complicated (pp. 19-38). Washington, DC: American Psychological Association.
Llabre, M. M., Spitzer, S. B., Saab, P. G., Ironson, G. H., & Schneiderman, N. (1991). The Reliability and Specificity of Delta Versus Residualized Change as Measures of Cardiovascular Reactivity to Behavioral Challenges. Psychophysiology, 28(6), 701 - 711.
Locke, K. D. (2000). Circumplex scales of interpersonal values: Reliability, validity, and applicability to interpersonal problems and personality disorders. Journal of Personality Assessment, 75(2), 249-267.
Matthews, K., Gallo, L. C., & Taylor, S. E. (2010). Are psychosocial factors mediators of socioeconomic satus and health connections? A progress report and blueprint for the future. Annals of the New York Acadamy of Sciences, 1186, 146-173.
Mendelson, T., Thurston, R. C., & Kubzansky, L. D. (2008). Affective and cardiovascular effects of experimentally-induced social status. Health Psychology, 27(4), 482-489.
Mendes, W. B., Blascovich, I., Major, B., & Seery, M. D. (2001). Challenge and threat responses during downward and upward social comparisons. European Journal of Social Psychology, 31, 477-497.
Miller, R., Plessow, F., Rauh, M., Groschl, M., & Kirschbaum, C. (2013). Comparison of salivary cortisol as measured by different immunoassays and tandem mass spectrometry. Psychoneuroendocrinology, 38, 50-57.
Nealey-Moore, J. B., Smith, T. W., Uchino, B. N., Hawkins, M. W., & Olson-Cerny, C. (2007). Cardiovascular reactivity during positive and negative marital interactions. Journal of Behavioral Medicine, 30, 505-519.
Newton, T. L. (2009). Cardiovascular functioning, personality, and the social world: the domain of hierarchical power. Neuroscience and Biobehavioral Reviews, 33(2), 145-159.
Operario, D., Adler, N. E., & Williams, D. R. (2004). Subjective social status: Reliability and predictive utility for global health. Psychology & Health, 19, 237-246.
Scheepers, D. (2009). Turning social identity threat into challenge: Status stability and cardiovascular reactivity during intergroup competition. Journal of Experimental Social Psychology, 45, 228 - 233.
Schmidt, J. A., Wagner, C. C., & Kiesler, D. J. (1999). Psychometric and circumplex properties of the octant scale Impact Message Inventory (IMI-C): A structural evaluation. Journal of Counseling Psychology, 46(3), 325-334.
Smith, T.W., & Christensen, A.J. (1992). Cardiovascular reactivity and interpersonal relations: Psychosomatic processes in social context. Journal of Social and Clinical Psychology, 11, 279-301.
Smith, T. W., Cundiff, J. M., & Uchino, B. N. (2012). Interpersonal motives and cardiovascular response: Mechanisms linking dominance and social status with cardiovascular disease. In R. A. Wright & G. H. E. Gendolla (Eds.), How motivation affects cardiovascular response: Mechanisms and applications. (pp. 287-305). Washington, DC US: American Psychological Association.
Smith, T. W., Ruiz, J. M., & Uchino, B. N. (2000). Vigilance, active coping, and cardiovascular reactivity during social interaction in young men. Health Psychology, 19(4), 382-392.
Steptoe, A., & Kivimaki, M. (2013). Stress and cardiovascular disease: An update on current knowledge. Annual Review of Public Health, 34, 337-354.
Spielberger, C. D. (1980). Preliminary manual for the State-Trait Personality Inventory. Tampa: University of South Florida, Human Resources Institute.
Thayer, J. F., Hansen, A. L., & Johnson, B. D. (2008). Non-invasive assessment of autonomic influences on the heart: Impedance cardiography and heart rate variability. In L. Leuken & L. Gallo (Eds.), Handbook of physiological research methods in health psychology (pp. 183-209). Thousand Oaks, CA: Sage.
Wright, R. A., Killebrew, K., & Pimpalapure, D. (2002). Cardiovascular incentive effects where a challenge is unfixed: Demonstrations involving social evaluation, evaluator status, and monetary reward. Psychophysiology, 39(2), 188-197.